Protons and Electrons
|
PROTONS determine the identity of an element.
Different elements have different numbers of protons. The number of protons in an element is shown by the ATOMIC NUMBER. Hydrogen has 1 proton Helium has 2 protons Oxygen has 8 protons |
ELECTRONS determine the behavior of an element.
Different elements have different numbers of electrons. The number of electrons is the SAME as the number of protons. Hydrogen has 1 electron Helium has 2 electrons Oxygen has 8 electrons |
REMEMBER
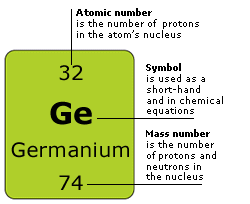
ATOMIC NUMBER = NUMBER OF PROTONS = NUMBER OF ELECTRONS
A = P = E
MASS NUMBER - ATOMIC NUMBER = NUMBER OF NEUTRONS
M - A = N
ATOMIC NUMBER = NUMBER OF PROTONS = NUMBER OF ELECTRONS
A = P = E
MASS NUMBER - ATOMIC NUMBER = NUMBER OF NEUTRONS
M - A = N
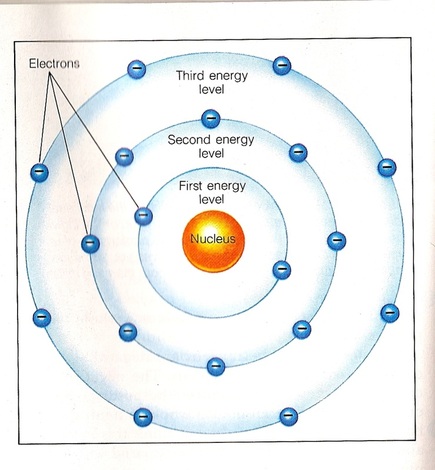
WHERE EXACTLY ARE THE ELECTRONS?
|
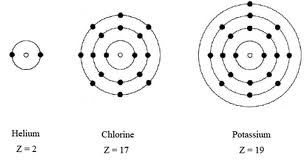
Electrons orbit around the nucleus in the electron cloud in a manner similar to the orbits of the planets around the sun. The orbit of the electrons are called ELECTRON SHELLS or ENERGY LEVELS. There is a maximum number of electrons that can fit in each shell. The electrons will fill up the shells starting at the innermost one first and then filling the next electron shell until all the electrons are used up. The electrons in the outermost shell are called VALENCE ELECTRONS Only 2 electrons can fit in the first electron shell (the one closest to the nucleus) Only 8 electrons can fit in the second and third shell |
|
Check your understanding!
|