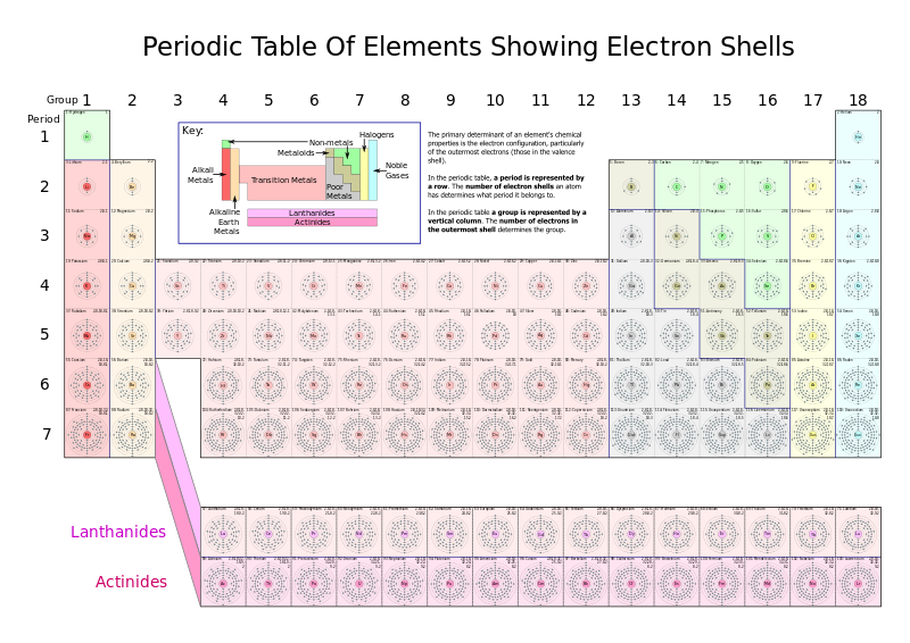
The Periodic table
REMEMEBER:
- Elements are arranged by increasing atomic mass
- The horizontal rows are called PERIODS. This tells you the number of ELECTRON SHELLS for an atom of an element in that row.
- The vertical columns are called GROUPS. This tells you the number of VALENCE ELECTRONS for an atom of an element in that column.
Check your understanding!
- Count the number of electron shells for every element in Period 1, 2, 3, and 4. Are the the same or are they different?
- Count the number of valence electrons for every element in Group 1, 2, 17, and 18. Are they the same or are they different?
- Do elements lose or gain electrons in Group 1 and 2 in order to become stable? What sort of ion do they become?
- Do elements lose or gain electrons in Groups 17 in order to become stable? What sort of ion to they become?